AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Name of bohr's atomic model3/18/2024  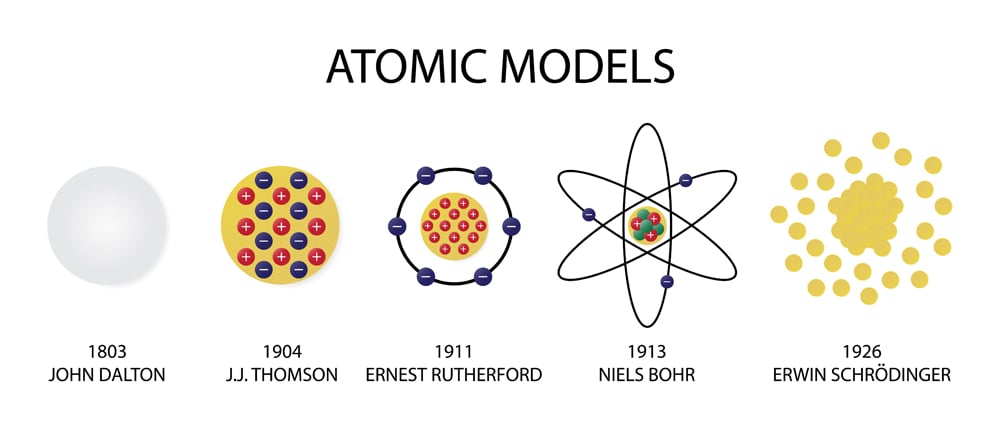
This idea was totally counter-intuitive and required a new physics. This is the same assumption as in Rutherford’s model and the centripetal force necessary for the circular motion is provided by the electrostatic force of attraction between the electron and the nucleus. The releasing of the photons is responsible for the line spectra given off by elements when excited. The postulates of Bohrs atomic model (for the hydrogen atom): The electrons revolve around the nucleus in circular orbits. Bohr’s atomic model proposed that atoms contain orbits that electrons travel along. Rutherford did not explain the relation of the orbital. A few remarkable applications of the Bohr model are as follows: The model has some significant impacts on setting up the grounds for quantum mechanics in explaining various observations. Bohr’s atomic theory was based on the Hydrogen line spectrum, while Rutherford’s atomic theory was on Gold foil. Even though the Bohr model is one of the simplest atomic models, it has many important applications.  
Then the electrons would release the photons in quantized amounts to return to ground state. Postulates were given in the year Niels Bohr in 1922, Ernest Rutherford in 1913. Why?īohr took the ideas of Rutherford (nuclear model), Planck (quanta), Einstein (photoelectric effect and spectroscopy and created the Planetary Model.īohr proposed that electrons were in energy levels (ground state) and absorbed photons of certain frequencies to move to a higher energy level (excited state). Classical physics says that opposites attract and likes repel, so the negative electrons should be attracted to the positive nucleus. Niels Bohr change the atomic theory by realizing that the electrons did not crash into the nucleus as would be expected in classical physics. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
